Loaʻa i ka hui Cyano ka polarity ikaika a me ka omo ʻana o ka electron, no laila hiki iā ia ke komo hohonu i loko o ka protein target e hana i nā pilina hydrogen me nā koena amino acid koʻikoʻi ma kahi hana. I ka manawa like, ʻo ka hui cyano ke kino isosteric bioelectronic o carbonyl, halogen a me nā hui hana ʻē aʻe, hiki ke hoʻoikaika i ka pilina ma waena o nā molekala lāʻau liʻiliʻi a me nā protein target, no laila ua hoʻohana nui ʻia i ka hoʻololi ʻana i ke ʻano o ka lāʻau lapaʻau a me nā pesticides [1]. ʻO nā lāʻau lapaʻau cyano e loaʻa ana i ka saxagliptin (Kiʻi 1), verapamil, febuxostat, etc; ʻO nā lāʻau lapaʻau mahiʻai e komo pū me ka bromofenitrile, fipronil, fipronil a pēlā aku. Eia kekahi, he waiwai nui ko nā hui cyano i nā kahua o ka ʻala, nā mea hana a pēlā aku. No ka laʻana, ʻo Citronitrile kahi ʻala nitrile hou o ka honua, a ʻo 4-bromo-2,6-difluorobenzonitrile kahi mea maka koʻikoʻi no ka hoʻomākaukau ʻana i nā mea kristal wai. Hiki ke ʻike ʻia ua hoʻohana nui ʻia nā hui cyano ma nā kahua like ʻole ma muli o ko lākou mau waiwai kū hoʻokahi [2].
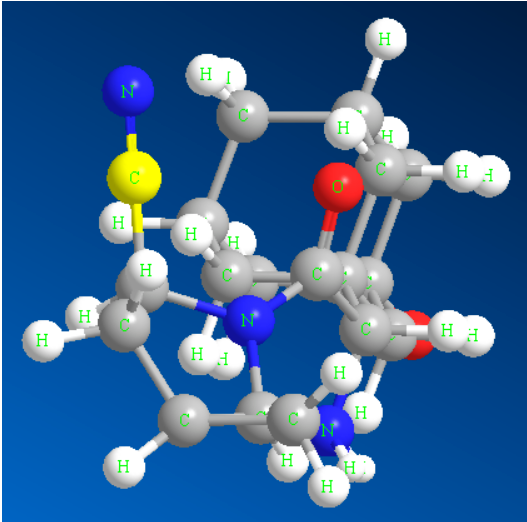
Loaʻa i ka hui Cyano ka polarity ikaika a me ka omo ʻana o ka electron, no laila hiki iā ia ke komo hohonu i loko o ka protein target e hana i nā pilina hydrogen me nā koena amino acid koʻikoʻi ma kahi hana. I ka manawa like, ʻo ka hui cyano ke kino isosteric bioelectronic o carbonyl, halogen a me nā hui hana ʻē aʻe, hiki ke hoʻoikaika i ka pilina ma waena o nā molekala lāʻau liʻiliʻi a me nā protein target, no laila ua hoʻohana nui ʻia i ka hoʻololi ʻana i ke ʻano o ka lāʻau lapaʻau a me nā pesticides [1]. ʻO nā lāʻau lapaʻau cyano e loaʻa ana i ka saxagliptin (Kiʻi 1), verapamil, febuxostat, etc; ʻO nā lāʻau lapaʻau mahiʻai e komo pū me ka bromofenitrile, fipronil, fipronil a pēlā aku. Eia kekahi, he waiwai nui ko nā hui cyano i nā kahua o ka ʻala, nā mea hana a pēlā aku. No ka laʻana, ʻo Citronitrile kahi ʻala nitrile hou o ka honua, a ʻo 4-bromo-2,6-difluorobenzonitrile kahi mea maka koʻikoʻi no ka hoʻomākaukau ʻana i nā mea kristal wai. Hiki ke ʻike ʻia ua hoʻohana nui ʻia nā hui cyano ma nā kahua like ʻole ma muli o ko lākou mau waiwai kū hoʻokahi [2].
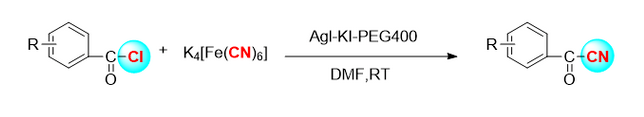
2.2 ka hopena cyanidation electrophilic o ka enol boride
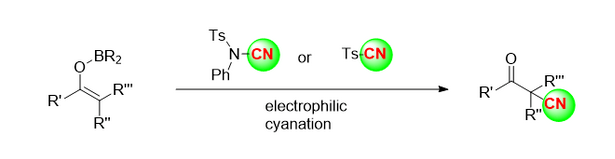
Ua hoʻohana ka hui o Kensuke Kiyokawa [4] i nā reagents cyanide n-cyano-n-phenyl-p-toluenesulfonamide (NCTS) a me p-toluenesulfonyl cyanide (tscn) e hoʻokō i ka cyanidation electrophilic kiʻekiʻe o nā hui enol boron (Kiʻi 3). Ma o kēia hoʻolālā hou, nā β-Acetonitrile like ʻole, a he ākea ka laulā o nā substrates.
2.3 ka hopena silico cyanide stereoselective catalytic organik o nā ketones
I kēia mau lā iho nei, ua hōʻike ka hui papa inoa ʻo Benjamin [5] ma ka puke pai ʻo Nature i ka hoʻokaʻawale ʻana o ka enantiomeric o 2-butanone (Kiʻi 4a) a me ka hopena cyanide asymmetric o 2-butanone me nā enzymes, nā mea hoʻouluulu organik a me nā mea hoʻouluulu metala hoʻololi, me ka hoʻohana ʻana iā HCN a i ʻole tmscn ma ke ʻano he reagent cyanide (Kiʻi 4b). Me ka tmscn ma ke ʻano he reagent cyanide, ua hoʻokau ʻia ʻo 2-butanone a me kahi ākea o nā ketones ʻē aʻe i nā hopena silyl cyanide enantioselective kiʻekiʻe ma lalo o nā kūlana catalytic o idpi (Kiʻi 4C).
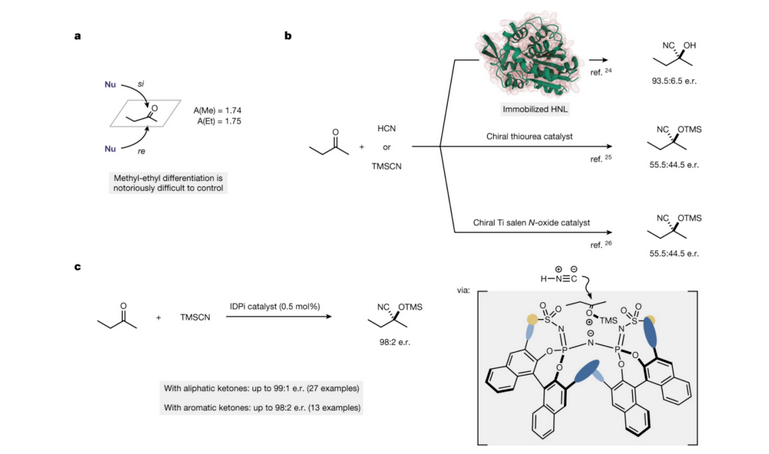
Kiʻi 4 A, ka hoʻokaʻawale ʻana o ka enantiomeric o 2-butanone. b. ʻO ka cyanidation asymmetric o 2-butanone me nā enzymes, nā mea hoʻouluulu organik a me nā mea hoʻouluulu metala hoʻololi.
c. Hoʻoulu ʻo Idpi i ka hopena silyl cyanide enantioselective kiʻekiʻe o 2-butanone a me kahi ākea o nā ketones ʻē aʻe.
2.4 ka cyanidation hoʻemi o nā aldehydes
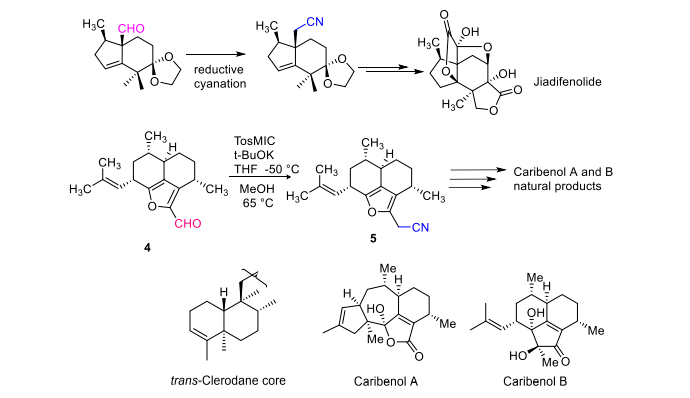
I ka hana ʻana o nā huahana kūlohelohe, hoʻohana ʻia ka tosmic ʻōmaʻomaʻo ma ke ʻano he reagent cyanide e hoʻololi maʻalahi i nā aldehydes i hoʻopaʻa ʻia i loko o nā nitriles. Hoʻohana hou ʻia kēia ʻano hana e hoʻokomo i kahi ʻātoma kalapona hou i loko o nā aldehydes a me nā ketones. He mea nui kēia ʻano hana i ka synthesis Enantiospecific holoʻokoʻa o jiadifenolide a he ʻanuʻu koʻikoʻi ia i ka synthesis o nā huahana kūlohelohe, e like me ka synthesis o nā huahana kūlohelohe e like me clerodane, caribenol A a me caribenol B [6] (Kiʻi 5).
2.5 ka hopena cyanide electrochemical o ka amine organik
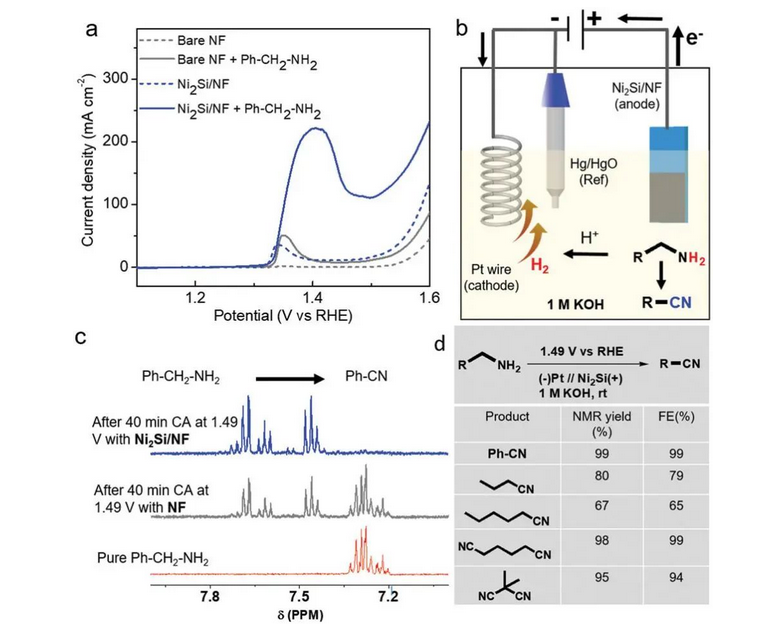
Ma ke ʻano he ʻenehana synthesis ʻōmaʻomaʻo, ua hoʻohana nui ʻia ka synthesis electrochemical organik ma nā ʻano like ʻole o ka synthesis organik. I nā makahiki i hala iho nei, ua nui aʻe nā mea noiʻi i nānā iā ia. Ua hōʻike koke aku nei ʻo PrashanthW. Ua hōʻike ʻo Menezes team [7] hiki ke hoʻopili pololei ʻia ka amine aromatic a i ʻole ka amine aliphatic i nā hui cyano like i loko o ka hopena 1m KOH (me ka ʻole o ka hoʻohui ʻana i ka reagent cyanide) me kahi hiki mau o 1.49vrhe me ka hoʻohana ʻana i ka mea hoʻoulu Ni2Si haʻahaʻa, me ka hua kiʻekiʻe (Kiʻi 6).
hōʻuluʻulu manaʻo 03
He hopena synthesis organik koʻikoʻi loa ka Cyanidation. Hoʻomaka mai ka manaʻo o ka kemika ʻōmaʻomaʻo, hoʻohana ʻia nā reagents cyanide aloha kaiapuni e pani i nā reagents cyanide ʻawahia a ʻino kuʻuna, a hoʻohana ʻia nā ʻano hana hou e like me ka solvent-free, non catalytic a me ka microwave irradiation e hoʻonui hou aku i ka laulā a me ka hohonu o ka noiʻi, i mea e loaʻa ai nā pono waiwai nui, pilikanaka a me ke kaiapuni i ka hana ʻoihana [8]. Me ka holomua mau o ka noiʻi ʻepekema, e ulu ka hopena cyanide i ka hua kiʻekiʻe, ka hoʻokele waiwai a me ka kemika ʻōmaʻomaʻo.
Ka manawa hoʻouna: Sep-07-2022